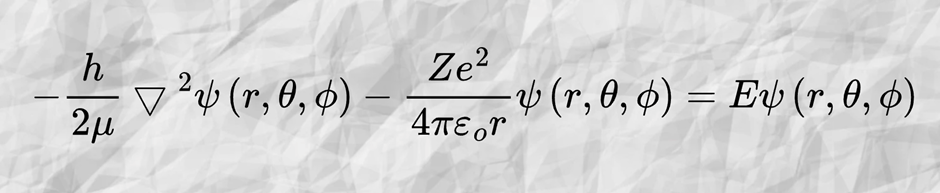You already know that electron is very small and assume to be a particle. In that microscopic world, the rule of Newton’s physics that we use for throwing a ball does not applied. The elctrons will likely as a “ghost” or waves, because we cannot know where exactly they are. This world we call as the quantum physics.
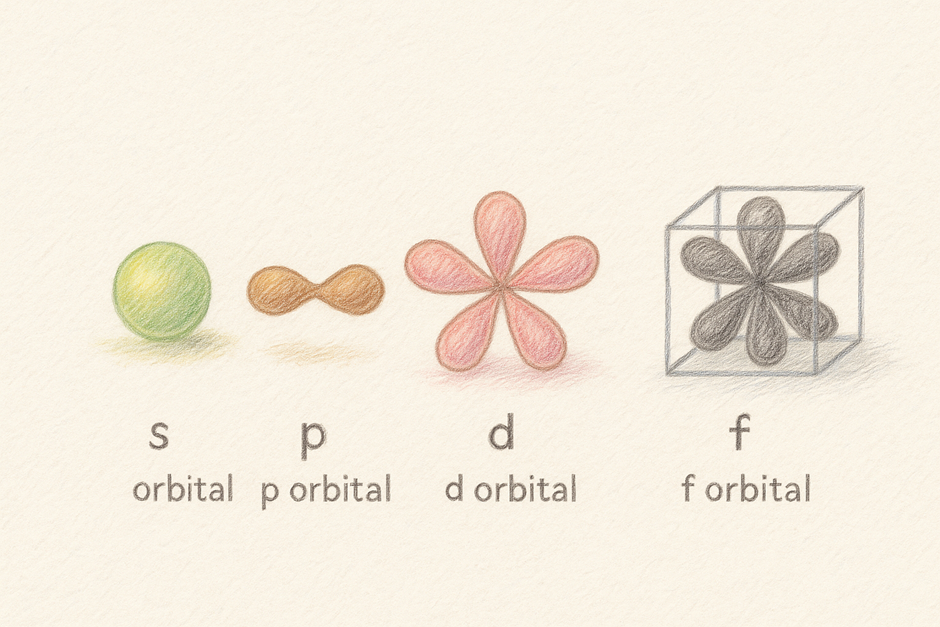
In chemistry, you learn about the shape of orbital: where the ‘s’ orbital with the spherical shape, the ‘p’ orbital looks like a dumbell on three directions perpendicular to each other, and the other orbitals speak for themselves. You have been told that this is the probability to find an electon, but what exactly does that means? And why and how the orbital shape determined.
The orbital shape is actually related to both quantum physics and chemistry. Even though some of you have not yet learned the equation in detail, but you can imagine this is the mathematical “recipe” to imagine how an electron’s wave behaves near the nucleus. Let us call the equation as the wavefunction, ψ, and it will be our map of probabilities.
The wave function ψ is not the orbital itself, but when we do some calculation, it gives us the number of probabilities to find the electron at that point. The greater the value of the calculation, the greater the probabilities to find the electron there. By solving the famous Schrodinger equation to get the wave function ψ, by considering the electron energy and its potential, we are going to plot the equation |wave function| 2, |ψ|2. This plotting will give the shape of the orbital.
From here we can see that when we plot for the lowest energy, which you learned in phsyics which is the ground state with energy of E=-13.6 eV, it gaves us a spherical shape, named as the ‘s’ orbital. Then, for the ‘p’ orbital, it depends on the direction. The plotting gives us the shape of dumbell with its center on the nucleus and in three axes: x, y and z. This difference in axis is due to the condition of magnetic quantum number restrictions that we put in when solving the Schrodinger equation.
This shape just shows the probabilities of finding an electron in those energy, not how its orbits the nucleus. We can say that you can find the electron anywhere inside the orbital and it is imposible to find outside the orbital. Why this is not the path of an electron orbiting the nucleus? This question you may answer when you choose to dive more deeply in the quantum world, and I hope it you do too.

by: Nor Syazana binti Shamsuddin
Date of Input: 06/05/2025 | Updated: 05/06/2025 | hasniah
MEDIA SHARING